Pikes Peak Ethics Committee Meeting
- First Friday of every month at noon.
- Email me if you would like to be added to our email list to receive the agenda and reminders about the meeting.
- https://zoom.us/j/97461641616?pwd=YPpJU1ZHMG3bTChSVxAcD9gb39qVaY.1
Innovage Program
- comfort care program
- This will be discussed at ethics committee meeting in December
Skin Substitutes (beware!) – Proposed CMDA position statement DRAFT
- Bioengineered Skin Substitutes – The Short version
- These are very expensive, overused and companies that make them are very aggressively pushing their use.
- CMS is working to line up the use of these skin substitutes and reimbursement with the evidence base.
- These substitutes have a very limited evidence-based role for use in LTC wound care.
- If anyone approaches you encouraging, you to use these skin substitutes…. let’s discuss it first. discuss it.
Based on a review of the literature and input from clinical experts, we have concluded that
bioengineered skin substitutes are rarely appropriate in long-term care settings. While they
may offer benefit in specific cases—such as diabetic foot or venous leg ulcers in patients meeting
defined criteria—their broad use across wound types is not supported by current evidence and
does not reflect the standard of care.
RE: Use of Bioengineered Skin Substitutes (AKA Allografts)
Bioengineered skin substitutes—often derived from human or animal tissue—have become
increasingly common in wound care, yet evidence supporting their use remains limited, and
these products are not subject to full FDA approval.
Across our region, provider groups have observed inappropriate and unsafe use of these
treatments in long-term care patients. Outcomes associated with use have included wound
deterioration, infections, hospitalizations, amputations, and even a death.
We support the recent action by the Centers for Medicare & Medicaid Services (CMS), which
reclassifies many of these products as “incident-to” supplies starting in 2026. This change
reflects growing concern that these treatments are too often used without clear clinical
benefit, putting patients at risk for complications without improving outcomes. The CMS
decision is a step toward restoring appropriate, patient-centered wound care by reinforcing
the importance of evidence-based treatment.
Based on a review of the literature and input from clinical experts, we have concluded that
bioengineered skin substitutes are rarely appropriate in long-term care settings. While they
may offer benefit in specific cases—such as diabetic foot or venous leg ulcers in patients meeting
defined criteria—their broad use across wound types is not supported by current evidence and
does not reflect the standard of care.
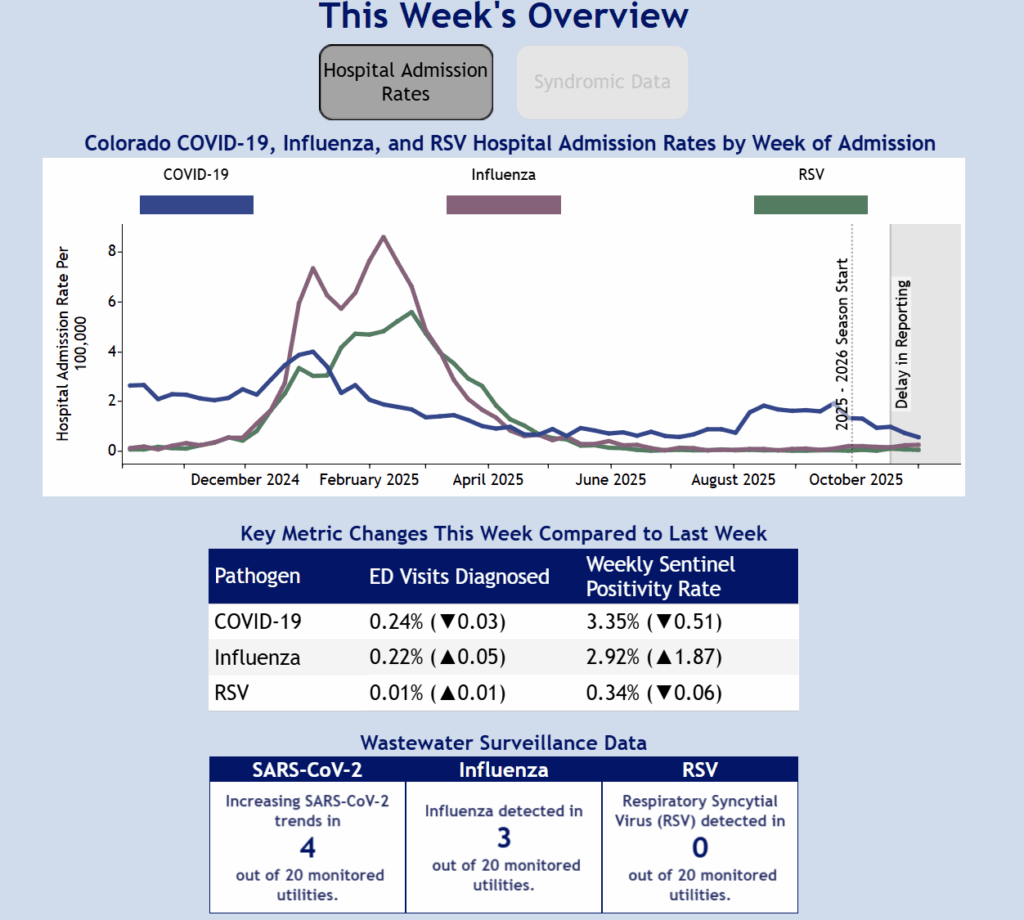
Influenza and COVID
- This is the month to give influenza and COVID vaccines.
Risks of Tramadol Likely Outweigh Benefit for Chronic Pain

https://www.medpagetoday.com/neurology/painmanagement/117842
Conclusion Tramadol may have a slight effect on reducing chronic pain levels (low certainty of evidence) while likely increasing the risk of both serious (moderate certainty of evidence) and non-serious adverse events (very low certainty of evidence). The potential harms associated with tramadol use for pain management likely outweigh its limited benefits.
