Things We Do for No Reason: Prescribing gabapentinoids for pain
Journal of Hospital Medicine: Things We Do for No Reason- Full Article
What are gabapentinoids?
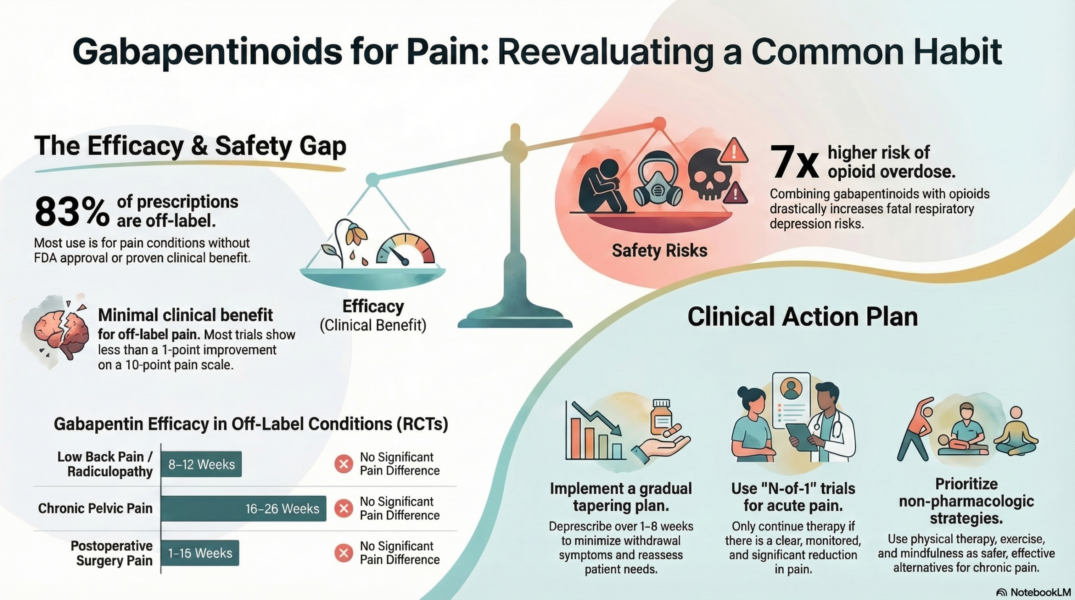
- Gabapentin and pregabalin — among the most prescribed drugs in the US (70M and 6.5M prescriptions in 2021, respectively)
- FDA-approved only for select conditions (postherpetic neuralgia, diabetic peripheral neuropathy, fibromyalgia, seizures)
- ~83% of prescriptions are off-label
The evidence against routine use:
- RCTs consistently show minimal or no benefit over placebo for most off-label pain conditions (back pain, sciatica, pelvic pain, postoperative pain, etc.)
- When benefits were found, they were often clinically insignificant (<1 point on a 0–10 pain scale)
- Pfizer paid billions in legal settlements for illegally promoting off-label use and suppressing negative trial data
Key harms and risks:
- Dizziness and sedation in up to one-third of users
- 7x increased risk of opioid overdose when combined with opioids
- Falls, delirium, respiratory depression, fractures
- Higher hospitalization risk, even at low doses
- Misuse and dependence, even at therapeutic doses
- Dangerous in renal impairment (renally cleared), yet dosing is often not adjusted
When they might still be considered:
- Postherpetic neuralgia and diabetic peripheral neuropathy (FDA-approved indications)
- Individualized “N-of-1” trials with clear goals, monitoring, and a plan to stop if ineffective
Recommendations:
- Don’t routinely prescribe gabapentinoids for pain
- Reassess at discharge and deprescribe when appropriate (taper over 1–2 weeks, or 4–8 weeks for long-term users)
- Prefer non-pharmacologic alternatives: physical therapy, cognitive-behavioral therapy, mindfulness, exercise